It catalyzes the removal of nearly any amino acid from the carboxyl end of any peptide or protein. The enzyme carboxypeptidase, on the other hand, is far less specific. Urease, for example, is an enzyme that catalyzes the hydrolysis of a single substrate-urea-but not the closely related compounds methyl urea, thiourea, or biuret. Some enzymes even distinguish between D- and L-stereoisomers, binding one stereoisomer but not the other. Some enzymes act on a single substrate, while other enzymes act on any of a group of related molecules containing a similar functional group or chemical bond. 
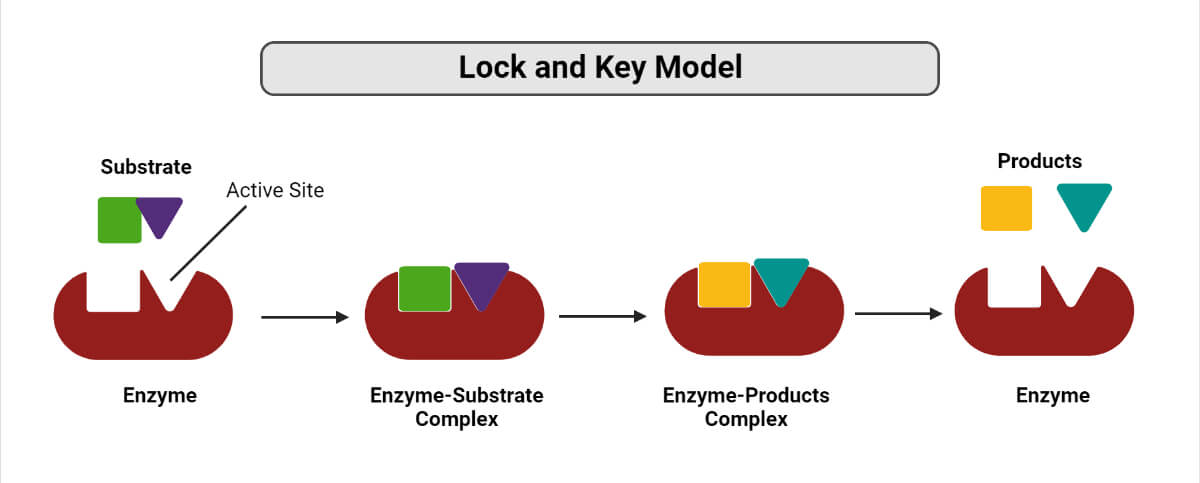
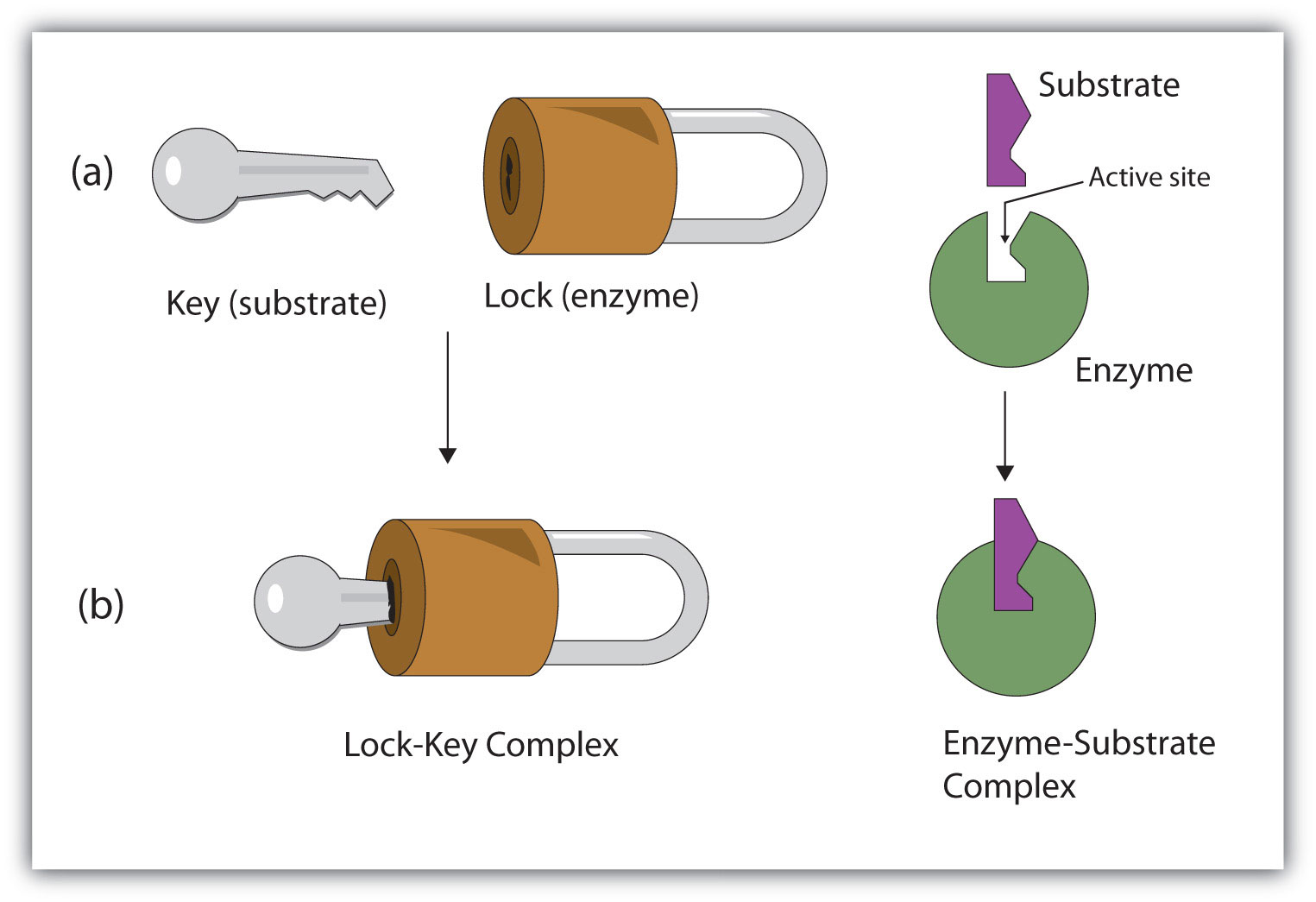
In contrast, enzymes are much more specific. Only the correct size key, which is the substrate, enters the keyhole, which is the active. An inorganic acid such as sulfuric acid can be used to increase the reaction rates of many different reactions, such as the hydrolysis of disaccharides, polysaccharides, lipids, and proteins, with complete impartiality. In this case, the enzyme is the lock, and the substrate is the key. One characteristic that distinguishes an enzyme from all other types of catalysts is its substrate specificity. Suggest an amino acid whose side chain might be in the active site of an enzyme and form the type of interaction you just identified.What type of interaction would occur between an COO − group present on a substrate molecule and a functional group in the active site of an enzyme?.Binding to enzymes brings reactants close to each other and aligns them properly, which has the same effect as increasing the concentration of the reacting compounds. They must bind to a specific substrate before they can catalyze a chemical reaction. The participating amino acids, which are usually widely separated in the primary sequence of the protein, are brought close together in the active site as a result of the folding and bending of the polypeptide chain or chains when the protein acquires its tertiary and quaternary structure. Lock-and-key model is a model for Substrate (chemistry) - Wikipedia interaction suggesting that the enzyme and the substrate possess specific complementary geometric shapes that fit exactly into one another. 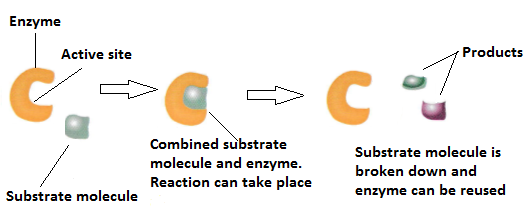
Amino acid side chains in or near the binding site can then act as acid or base catalysts, provide binding sites for the transfer of functional groups from one substrate to another or aid in the rearrangement of a substrate. The structural changes that occur when an enzyme and a substrate join together bring specific parts of a substrate into alignment with specific parts of the enzyme’s active site. This makes enzymes highly specific each type of enzyme. (b) The enzyme conformation changes dramatically when the substrate binds to it, resulting in additional interactions between hexokinase and glucose. In the lock and key model, the shape of the active site matches the shape of its substrate. In 1894, German chemist Emil Fischer proposed the lock and key theory, which states that enzymes have a specific shape that directly correlates to the shape of the substrate. (a) The enzyme hexokinase without its substrate (glucose, shown in red) is bound to the active site. However, it does not explain the stabilization of the transition state that the enzymes achieve.\): The Induced-Fit Model of Enzyme Action. The lock and key model theory first postulated by Emil Fischer in 1894 shows the high specificity of enzymes. Unlike the lock-and-key model, the induced fit model shows that enzymes are rather flexible structures. As for the induced fit model suggested by Daniel Koshland in 1958, it suggests that the active site continues to change until the substrate is completely bound to the active site of the enzyme, at which point the final shape and charge are determined. Like a key into a lock, only the correct size and shape of the substrate ( the key) would fit into the active site ( the key hole) of the enzyme ( the lock). In the lock-and-key model, the enzyme-substrate interaction suggests that the enzyme and the substrate possess specific complementary geometric shapes that fit exactly into one another. At present, there are two models, which attempt to explain enzyme specificity:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
